ShmoopTube
Where Monty Python meets your 10th grade teacher.
Search Thousands of Shmoop Videos
Test Prep Videos 443 videos
ACT Science: Research Summary Passage Drill 2, Problem 1. Why do you think that the filter paper will not remove the salt from the water?
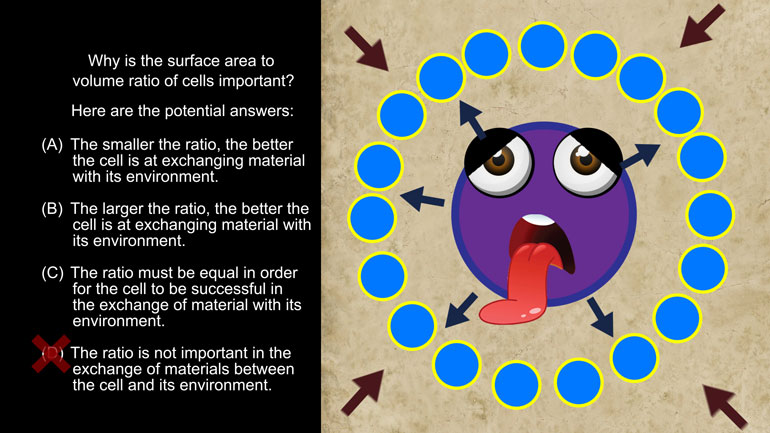
AP Biology: Biological System Interactions Drill 1, Problem 1. Complete the sentence about a saturated fatty acid.
AP Biology: Essential Life Process Information Drill 1, Problem 1. If one parent is heterozygous for the sickle cell trait while the other par...
AP Chemistry 2.5 Chemical Reaction Rates 10 Views
Share It!
Description:
Want to pull an Oliver Twist and ask us for more? We've gotcha covered. Head over to /video/subjects/test-prep/ap/ap-chemistry/ for more AP Chemistry goodness. Porridge not included.
Transcript
- 00:03
And here’s your Shmoop du jour, brought to you by forward reactions, when somebody
- 00:08
responds to your flirting with… more flirting. [Man gives woman a bunch of flowers]
- 00:10
All right, here’s today’s question:
- 00:12
Which of the following is true of catalysts?
- 00:15
And here are your potential answers: All right, let’s get up to speed on catalysts. [Men cycling]
Full Transcript
- 00:22
The purpose of a catalyst is to increase the rate of a reaction.
- 00:26
Meaning that a substance that inhibits a reaction is not a catalyst… [Catalyst added to a beaker]
- 00:29
Duh.
- 00:30
So we can cross out D right off the bat.
- 00:33
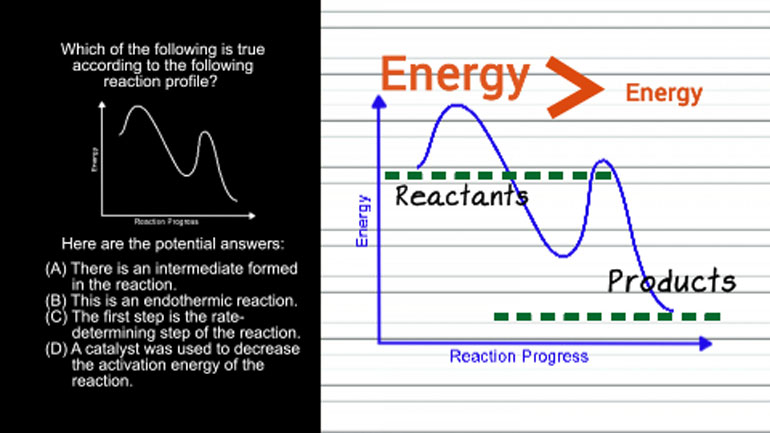
And how does a catalyst increase a reaction rate? [A reaction rate graph]
- 00:36
By lowering the energy of an intermediate or transition state in the reaction mechanism.
- 00:41
This decreases the activation energy barrier, which makes the reaction go faster.
- 00:45
If only there were catalysts for doing that 8-page paper that’s due tomorrow, eh? [Woman scientist staring at a catalyst]
- 00:50
Just like your car, chemical reactions can go in the forward and reverse directions. [Car travelling up and down a hill]
- 00:54
The mechanism for the reverse direction is often just the forward mechanism, backwards.
- 00:58
That makes sense, right? [Car reverses up a hill]
- 01:00
So if a catalyst lowers the energy of a transition state, it can make both the forward and reverse
- 01:05
reactions faster.
- 01:07
That means that the correct answer is B. Now don’t go get that paper done. [Person orders boy to finish his paper]
- 01:12
That was reverse psychology.
- 01:14
Is it working? [Boy working on his paper at his desk]
Related Videos
AP Chemistry 1.3 Chemical Reaction Rates. What is the overall order of the reaction?
AP Chemistry 1.4 Chemical Reaction Rates. What are the correct units for a second order rate constant?
AP Chemistry 1.5 Chemical Reaction Rates. What is the rate law for the reaction?
AP Chemistry 3.2 Laws of Thermodynamics. What is the value for ΔG?